
Media
Disappointed by the FDA’s ruling on an investigational EPP treatment
Porphyria News: A patient-voice commentary piece reflecting on the FDA decision and why the community remains hopeful while facing the emotional reality of the delay.
Two local women share life with rare sun condition, urge continued drug development
Boston 25 News: Ginger Gonsalves and Becky Kelliher joined Kerry Kavanaugh to discuss their experience living with EPP and their positive experience on bitopertin, and to advocate for approval of bitopertin.
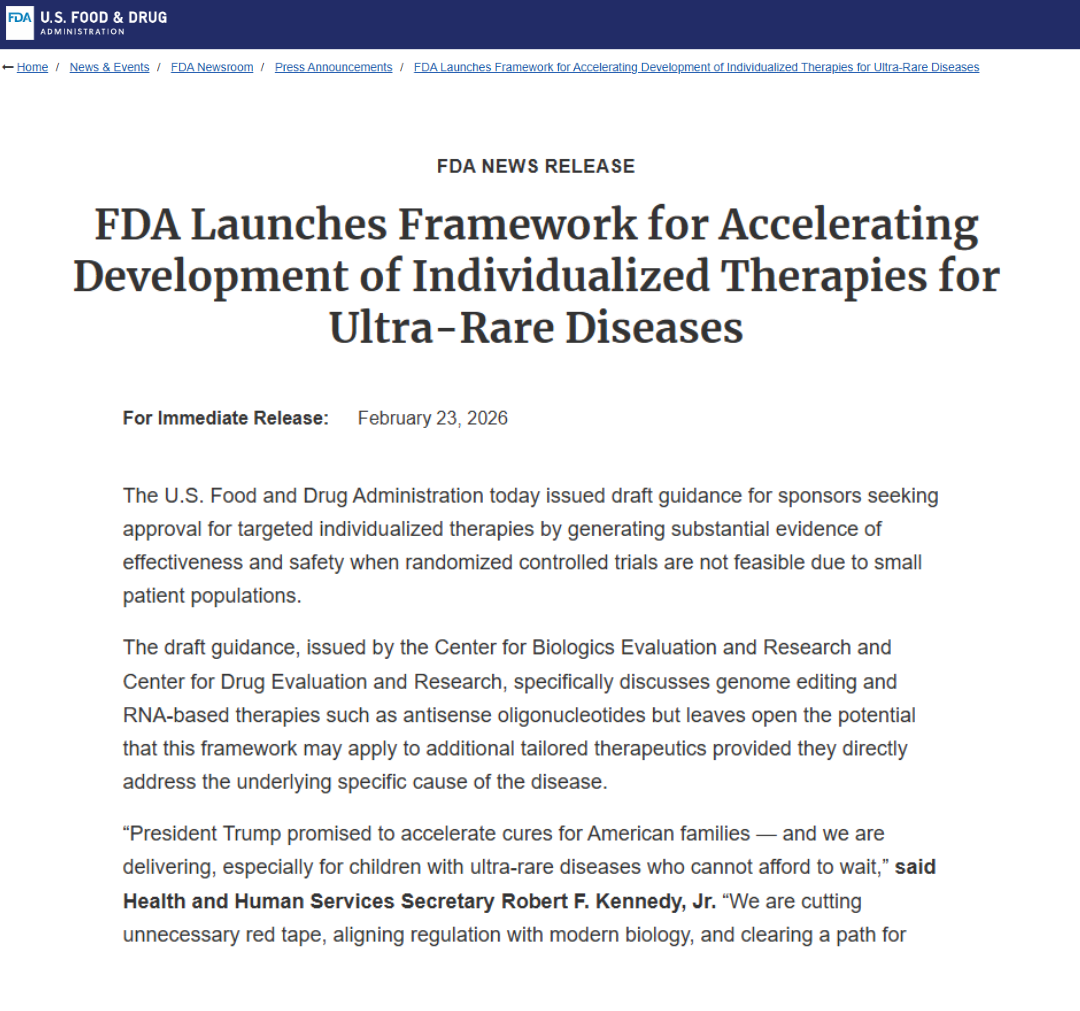
FDA Launches Framework for Accelerating Development of Individualized Therapies for Ultra-Rare Diseases
FDA Press Announcement: The FDA announced a new framework and draft guidance aimed at helping sponsors generate evidence of safety and effectiveness when randomized trials are not feasible because patient populations are extremely small. This is one of the clearest recent statements of FDA intent to create more workable pathways for ultra-rare disease therapies.
Delay for Disc as CNPV-designated bitopertin earns CRL in EPP
BioWorld: A piece focused on the regulatory angle, including questions about consistency and stability at the FDA after a CRL was issued for a drug that had been designated under the Commissioner’s National Priority Voucher pathway.
FDA rejects Disc’s rare disease drug despite Commissioner’s voucher
Fierce Biotech: This story zeroes in on the pathway issue, explaining that the FDA agreed the trial was adequate and well-controlled but objected to the surrogate endpoint and the lack of a sufficiently demonstrated link to clinical outcomes.
US FDA declines to approve Disc Medicine’s rare disease drug
Reuters: A clear overview of the CRL, including that bitopertin had been reviewed under the FDA’s Commissioner’s National Priority Voucher program and that the agency did not find enough evidence linking the biomarker response to clinical benefit.

He Grew Up Allergic to Sunlight. Now He Makes Wishes Come True for Kids with the Same Painful Condition
People: Craig Leppert shares his experience living with EPP, founding Shadow Jumpers, and what bitopertin has meant for people with this painful rare disease. The story helps bring the human side of EPP and treatment access into clear view.
FDA Rare Disease Innovation Hub Strategic Agenda
FDA Rare Disease Innovation Hub: The FDA Rare Disease Innovation Hub released its Strategic Agenda, outlining priorities for the year and emphasizing partnership with the rare disease community, regulatory innovation, and improved development pathways for rare disease products.
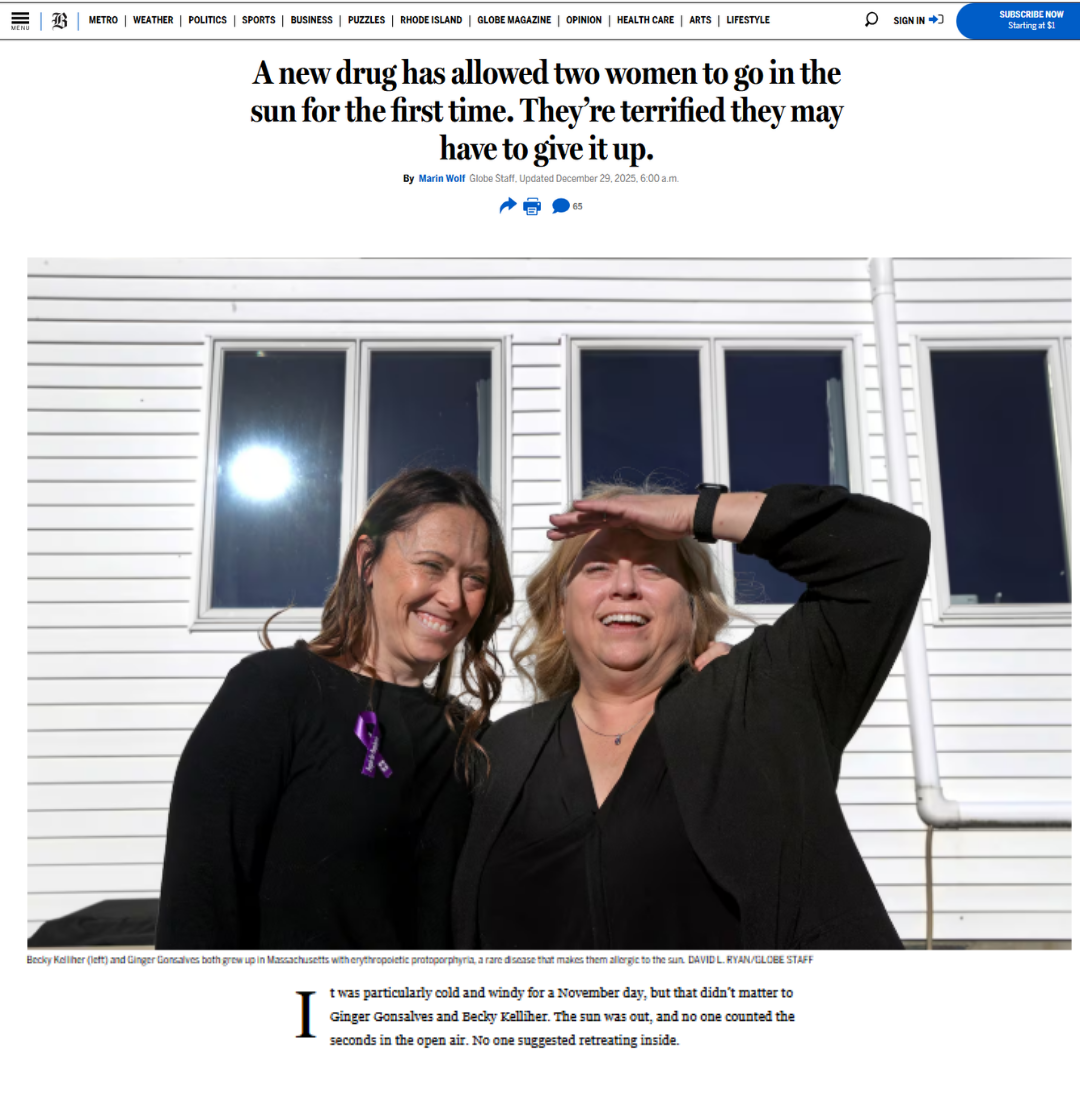
A new drug has allowed two women to go in the sun for the first time in years
The Boston Globe: A strong human-interest story that helps explain what bitopertin can mean in real life for people with EPP.
USD student diagnosed with rare sun disorder inspired to pursue biochemistry career
ABC 10News San Diego: This story features Sophia McClellan, a University of San Diego student from Southern California living with EPP. It focuses on how EPP shaped her life and academic path
FDA Advances Rare Disease Drug Development with New Evidence Principles
FDA Press Announcement: The FDA introduced the Rare Disease Evidence Principles to provide greater speed and predictability for therapies intended for very small rare disease populations with significant unmet need. The announcement specifically highlights clearer guidance on the kinds of evidence that may support approval in these settings.
